In this section, the activity coefficient models supported by the current version of SuperPro Designer are being presented.
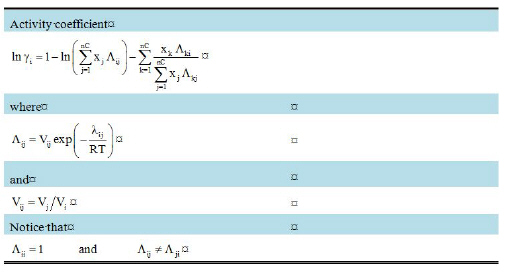
The Wilson model requires two interaction parameters λij and λji per binary pair. It accurately predicts multicomponent mixture equilibria. For aqueous solutions, NRTL is recommended over Wilson, because NRTL better predicts phase equilibrium properties. Wilson model can not be used to predict liquid-liquid equilibria.

The partial molar volumes Vi and Vj are typically approximated, with satisfactory accuracy, by the molar volumes of the pure substances i and j. In the absence of user provided values for λij and λji, zero values are used by SuperPro.
The NRTL model can be used to model vapor-liquid and liquid-liquid multi-component equilibrium systems. It performs better than Wilson for aqueous mixtures. The model requires two interaction parameters (gij, gji) and one binary parameter (aij).
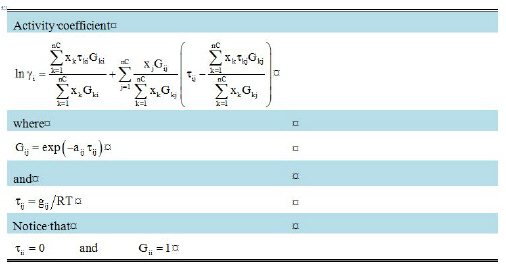
In the absence of user provided values gij gji and aij, zero values are used by SuperPro.
The SPT-NRTL (SMILEStoPropertiesTransformer NRTL) is a ten-parameter model that can be used to model the vapor-liquid equilibrium of mixtures with non-ionic components. It is implemented in SuperPro from the publication of Winter et al. in the Fluid Phase Equilibria journal. SPT-NRTL uses SMILES as molecular descriptors to predict activity coefficients. Specifically, the NRTL equation was integrated into a machine-learning model, trained with a very large set of experimental data, in order to estimate the required parameters. SPT-NRTL has eight “t” parameters (tij,1 to tji,4 and tji,1 to tji,4) and two “α” parameters (a1 and a2).
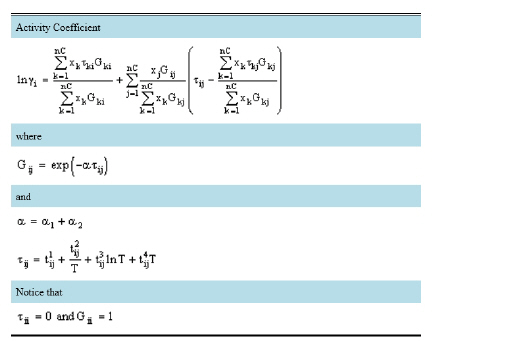
In the absence of user provided values for α1, α2 and τij, zero values are used by SuperPro.